21.3 Energy
A simple way to define energy is the ability to do work. Energy exists in different forms, including electrical energy, light energy, chemical energy, and heat energy.
Potential and Kinetic Energy
When an object is in motion, there is energy. For example, an airplane in flight produces considerable energy. This is because moving objects are capable of enacting a change, or doing work. Think of a wrecking ball. Even a slow-moving wrecking ball can do considerable damage to other objects. However, a wrecking ball that is not in motion is incapable of performing work. Energy with objects in motion is kinetic energy. A speeding bullet, a walking person, rapid molecule movement in the air (which produces heat), and electromagnetic radiation like light all have kinetic energy.
What if we lift that same motionless wrecking ball two stories above a car with a crane? If the suspended wrecking ball is unmoving, can we associate energy with it? The answer is yes. The suspended wrecking ball has associated energy that is fundamentally different from the kinetic energy of objects in motion. This energy form results from the potential for the wrecking ball to do work. If we release the ball it would do work. Because this energy type refers to the potential to do work, we call it potential energy. Objects transfer their energy between kinetic and potential in the following way: As the wrecking ball hangs motionless, it has 0 kinetic and 100 percent potential energy. Once it releases, its kinetic energy begins to increase because it builds speed due to gravity. Simultaneously, as it nears the ground, it loses potential energy. Somewhere mid-fall it has 50 percent kinetic and 50 percent potential energy. Just before it hits the ground, the ball has nearly lost its potential energy and has near-maximal kinetic energy. Other examples of potential energy include water’s energy held behind a dam, or a person about to skydive from an airplane.

The very existence of living cells relies heavily on potential energy. The bonds that hold molecules’ atoms together have potential energy. Remember that anabolic cellular pathways require energy to synthesize complex molecules from simpler ones, and catabolic pathways release energy when complex molecules break down. That the breakage of a chemical bond can release energy implies that those bonds have potential energy. In fact, there is potential energy stored within the bonds of all the food molecules we eat, which we eventually harness for use. This is because these bonds can release energy when broken. Scientists call the type of potential energy that exists within chemical bonds (that is released when those bonds break) chemical energy. Chemical energy is responsible for providing living cells with energy from food. Breaking the molecular bonds within fuel molecules brings about the energy’s release.
Free Energy
The amount of useful energy transferred during a chemical reaction is referred to as Gibbs free energy (abbreviated with the letter G) after Josiah Willard Gibbs, the scientist who developed the measurement. Recall that according to the second law of thermodynamics, all energy transfers involve losing some energy in an unusable form such as heat, resulting in entropy. Gibbs free energy specifically refers to the energy associated with a chemical reaction that is available after we account for entropy. In other words, Gibbs free energy refers to usable energy, or energy that is available to do work.
Every chemical reaction involves a change in free energy, called delta G (∆G). We can calculate the change in free energy for any system that undergoes such a change, such as a chemical reaction. To calculate ∆G, subtract the amount of energy lost to entropy (denoted as ∆S) from the system’s total energy change. The total energy in the system is enthalpy and we denote it as ∆H. The formula for calculating ∆G is as follows, where the symbol T refers to absolute temperature in Kelvin (degrees Celsius + 273):
∆G = ∆H – T∆S
We express a chemical reaction’s standard free energy change as an amount of energy per mole of the reaction product (either in kilojoules or kilocalories, kJ/mol or kcal/mol; 1 kJ = 0.239 kcal) under standard pH, temperature, and pressure conditions. We generally calculate standard pH, temperature, and pressure conditions at pH 7.0 in biological systems, 25 degrees Celsius, and 100 kilopascals (1 atm pressure), respectively. Note that cellular conditions vary considerably from these standard conditions, and so standard calculated ∆G values for biological reactions will be different inside the cell.
Endergonic Reactions and Exergonic Reactions
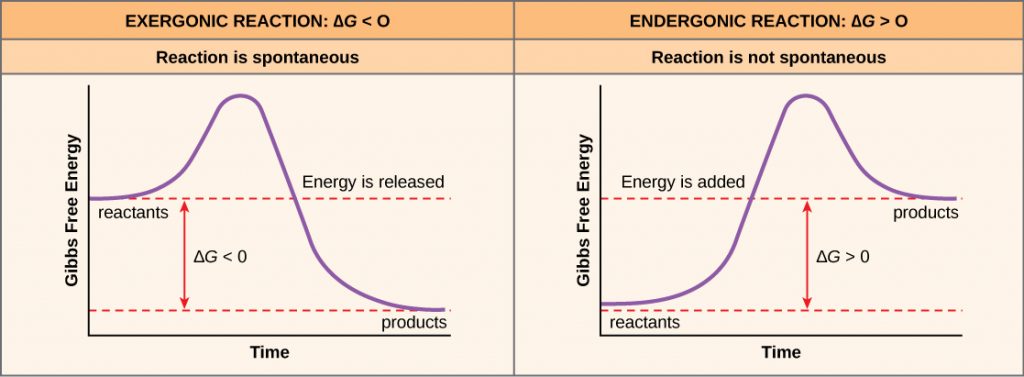
If energy is released during a chemical reaction, then the resulting value from the above equation will be a negative number. In other words, reactions that release energy have a ∆G < 0. A negative ∆G also means that the reaction’s products have less free energy than the reactants, because they gave off some free energy during the reaction. Scientists call reactions that have a negative ∆G and consequently release free energy exergonic reactions. Think: exergonic means energy is exiting the system. We also refer to these reactions as spontaneous reactions, because they can occur without adding energy into the system. Understanding which chemical reactions are spontaneous and release free energy is extremely useful for biologists, because these reactions can be harnessed to perform work inside the cell. We must draw an important distinction between the term spontaneous and the idea of a chemical reaction that occurs immediately. Contrary to the everyday use of the term, a spontaneous reaction is not one that suddenly or quickly occurs. Rusting iron is an example of a spontaneous reaction that occurs slowly, little by little, over time.
If a chemical reaction requires an energy input rather than releasing energy, then the ∆G for that reaction will be a positive value. In this case, the products have more free energy than the reactants. Thus, we can think of the reactions’ products as energy-storing molecules. We call these chemical reactions endergonic reactions, and they are non-spontaneous. An endergonic reaction will not take place on its own without adding free energy.
Most chemical reactions are reversible. They can proceed in both directions, releasing energy into their environment in one direction, and absorbing it from the environment in the other direction. For example, the breaking down of proteins releases energy and the building of proteins requires energy. Reactants within a closed system will undergo chemical reactions in both directions until they reach a state of equilibrium, which is the state with the lowest possible free energy and a state of maximal entropy. To push the reactants and products away from a state of equilibrium requires energy. Either reactants or products must be added, removed, or changed.
If a cell were a closed system, its chemical reactions would reach equilibrium, and it would die because there would be insufficient free energy remaining to perform the necessary work to maintain life. In a living cell, chemical reactions are constantly moving towards equilibrium, but never reach it. This is because a living cell is an open system. Materials pass in and out, the cell recycles the products of certain chemical reactions into other reactions, and there is never chemical equilibrium. In this way, living organisms are in a constant energy-requiring, uphill battle against equilibrium and entropy. This constant energy supply ultimately comes from sunlight.
the ability to do work
energy of objects or particles due to their motion
energy type that has the potential to do work; stored energy
potential energy in chemical bonds that releases when those bonds are broken
Gibbs free energy is the energy that is available to do work
the total energy of a system
describes chemical reactions that release free energy
describes chemical reactions that require energy input